Bile acids as modulators of gut microbiota composition and function (VSports app下载)
- PMID: 36740850
- PMCID: PMC9904317
- DOI: 10.1080/19490976.2023.2172671
V体育2025版 - Bile acids as modulators of gut microbiota composition and function
Abstract
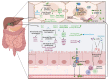
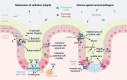
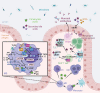
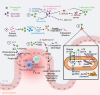
Changes in the composition of gut-associated microbial communities are associated with many human illnesses, but the factors driving dysbiosis remain incompletely understood. One factor governing the microbiota composition in the gut is bile. Bile acids shape the microbiota composition through their antimicrobial activity and by activating host signaling pathways that maintain gut homeostasis. Although bile acids are host-derived, their functions are integrally linked to bacterial metabolism, which shapes the composition of the intestinal bile acid pool. Conditions that change the size or composition of the bile acid pool can trigger alterations in the microbiota composition that exacerbate inflammation or favor infection with opportunistic pathogens VSports手机版. Therefore, manipulating the composition or size of the bile acid pool might be a promising strategy to remediate dysbiosis. .
Keywords: Bile acids; colonization resistance; intestinal homeostasis; microbiome. V体育安卓版.
Conflict of interest statement (VSports在线直播)
No potential conflict of interest was reported by the author(s).
Figures (V体育2025版)

References (V体育官网)
-
- Sender R, Fuchs S, Milo R.. Revised estimates for the number of human and bacteria cells in the body. PLoS Biol. 2016;14(8):e1002533. Available from: https://pubmed.ncbi.nlm.nih.gov/27541692/ - PMC - PubMed
-
- Levy M, Blacher E, Elinav E. Microbiome, metabolites and host immunity. Curr Opin Microbiol. 2017;35:8–27. Available from: https://pubmed.ncbi.nlm.nih.gov/27883933/. - PubMed
-
- Koh A, de Vadder F, Kovatcheva-Datchary P, Bäckhed F. From dietary fiber to host physiology: short-chain fatty acids as key bacterial metabolites. Cell [Internet] 2016; 165:1332–1345. Available from: http://www.ncbi.nlm.nih.gov/pubmed/27259147 - PubMed
-
- Louis P, Hold GL, Flint HJ. The gut microbiota, bacterial metabolites and colorectal cancer. Nat Rev Microbiol [Internet] 2014; 12:661–672. Available from: http://www.ncbi.nlm.nih.gov/pubmed/25198138. - PubMed
-
- McCarville JL, Chen GY, Cuevas VD, Troha K, Ayres JS. Microbiota metabolites in health and disease. Annu Rev Immunol [Internet] 2020; 38:147–170. Available from: http://www.ncbi.nlm.nih.gov/pubmed/32340573. - PubMed
Publication types
- Actions (V体育ios版)
MeSH terms
- "V体育2025版" Actions
- VSports - Actions
