Val-boroPro accelerates T cell priming via modulation of dendritic cell trafficking resulting in complete regression of established murine tumors (V体育官网入口)
- PMID: 23554941
- PMCID: PMC3595211
- DOI: 10.1371/journal.pone.0058860
"V体育安卓版" Val-boroPro accelerates T cell priming via modulation of dendritic cell trafficking resulting in complete regression of established murine tumors
Abstract
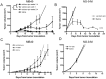
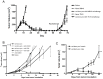
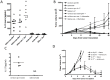
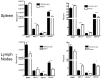
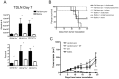
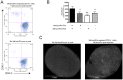
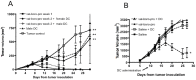
Although tumors naturally prime adaptive immune responses, tolerance may limit the capacity to control progression and can compromise effectiveness of immune-based therapies for cancer. Post-proline cleaving enzymes (PPCE) modulate protein function through N-terminal dipeptide cleavage and inhibition of these enzymes has been shown to have anti-tumor activity. We investigated the mechanism by which Val-boroPro, a boronic dipeptide that inhibits post-proline cleaving enzymes, mediates tumor regression and tested whether this agent could serve as a novel immune adjuvant to dendritic cell vaccines in two different murine syngeneic murine tumors VSports手机版. In mice challenged with MB49, which expresses the HY antigen complex, T cell responses primed by the tumor with and without Val-boroPro were measured using interferon gamma ELISPOT. Antibody depletion and gene-deficient mice were used to establish the immune cell subsets required for tumor regression. We demonstrate that Val-boroPro mediates tumor eradication by accelerating the expansion of tumor-specific T cells. Interestingly, T cells primed by tumor during Val-boroPro treatment demonstrate increased capacity to reject tumors following adoptive transfer without further treatment of the recipient. Val-boroPro -mediated tumor regression requires dendritic cells and is associated with enhanced trafficking of dendritic cells to tumor draining lymph nodes. Finally, dendritic cell vaccination combined with Val-boroPro treatment results in complete regression of established tumors. Our findings demonstrate that Val-boroPro has antitumor activity and a novel mechanism of action that involves more robust DC trafficking with earlier priming of T cells. Finally, we show that Val-boroPro has potent adjuvant properties resulting in an effective therapeutic vaccine. .
Conflict of interest statement
Figures



V体育官网 - References
-
- Vesely MD, Kershaw MH, Schreiber RD, Smyth MJ (2011) Natural Innate and Adaptive Immunity to Cancer. Annu Rev Immunol 29: 235–271. - PubMed (V体育平台登录)
-
- Koebel CM, Vermi W, Swann JB, Zerafa N, Rodig SJ, et al. (2007) Adaptive immunity maintains occult cancer in an equilibrium state. Nature 450: 903–907. - PubMed (V体育官网)
-
- Dunn GP, Bruce AT, Ikeda H, Old LJ, Schreiber RD (2002) Cancer immunoediting: from immunosurveillance to tumor escape. Nat Immunol 3: 991–998. - "V体育平台登录" PubMed
-
- Dunn GP, Old LJ, Schreiber RD (2004) The immunobiology of cancer immunosurveillance and immunoediting. Immunity 21: 137–148. - PubMed
-
- Khong HT, Restifo NP (2002) Natural selection of tumor variants in the generation of “tumor escape” phenotypes. Nat Immunol 3: 999–1005. - PMC (VSports手机版) - PubMed
Publication types
- "V体育平台登录" Actions
VSports app下载 - MeSH terms
- Actions (VSports手机版)
- VSports app下载 - Actions
- "VSports在线直播" Actions
- "VSports注册入口" Actions
- "V体育安卓版" Actions
- VSports - Actions
- "V体育2025版" Actions
- VSports最新版本 - Actions
- "VSports最新版本" Actions
- "VSports最新版本" Actions
- "VSports app下载" Actions
- "V体育安卓版" Actions
Substances
- "VSports" Actions
- Actions (VSports注册入口)
- VSports注册入口 - Actions
- "V体育官网" Actions
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
"VSports注册入口" Molecular Biology Databases

